-
Address:
17888 67th Court North
Loxahatchee, FL
-
Mail us:
contact@wrightacademia.org
- submit manuscript
Journal of Surgery and Clinical Reports
Table of content
Research Article |
Open Access |
Volume 1 | Issue 1 |
Short-and-Long-Term Outcomes of Hepatic Resection for Hepatocellular Carcinoma in Cirrhotic and Non-Cirrhotic Liver Parenchyma
Francisco Calderon, MD1, Esteban Masino, MD1, Lucas Caram, MD1, Victoria Ardiles, MD, PhD2, Jimena Vicens, MD3, Valeria Aliperti, MD3, Rodrigo Sánchez Claria, MD4, Oscar Mazza, MD4, Eduardo de Santibañes, MD, PhD2, Juan Pekolj, MD, PhD2 and Martín de Santibañes, MD, PhD2
1General Surgery, Hospital Italiano de Buenos Aires, Buenos Aires, Argentina
2Hepato-Pancreato-Biliary Surgery and Liver Transplantation Section, Hospital Italiano de Buenos Aires, Buenos Aires, Argentina
3Internal Medicine, Epidemiology Section, Hospital Italiano de Buenos Aires, Buenos Aires, Argentina
4Hepato-Pancreato-Biliary Surgery Section, Hospital Italiano de Buenos Aires, Buenos Aires, Argentina
*Corresponding author: MartÍn de Santibañes, MD, PhD, Department of Surgery, Division of HPB Surgery, Liver, Transplant Unit, Hospital Italiano de Buenos Aires, Juan D Perón 4190, C1181ACH. Buenos Aires, Argentina, Tel: +54-11-4981-4501, E-mail: martin.desantibanes@hospitalitaliano.org.ar
Citation: Calderon F, Masino E, Caram L, Ardiles V, Vicens J, et al. (2021) Short-and-Long-Term Outcomes of Hepatic Resection for Hepatocellular Carcinoma in Cirrhotic and Non-Cirrhotic Liver Parenchyma. J Surg Clin Rpts 1:003.
Copyright © Calderon F, et al.
Received: |
Accepted: |
Published: |
Background: Hepatocellular carcinoma (HCC) is the most frequent primary liver tumor, 90% of these setting in a cirrhotic liver. The aim of this study was to analyze the results of resected HCC in cirrhotic and non-cirrhotic patients in a 12 year period. Postoperative outcomes, overall survival (OS) and recurrence-free survival (RFS) were analyzed, as well factors involved.
Methods: The population consisted of patients with HCC who underwent surgical resection in our institution between January 2007 and December 2018. Data was retrospectively collected.
Results: 86 patients between 2007 and 2018 met inclusion criteria. 49 had a cirrhotic liver, while 37 patients had HCC in a non-cirrhotic liver. For cirrhotic patients, OS and RFS at 1, 3 and 5 years were 77%, 59%, 49% and 75.8%, 43.9%, 25%, respectively. Patients outside UCSF criteria (p = 0.05), several nodules (p = 0.004) and a larger tumor size (p = 0.02) were associated with poorer OS. Patients outside Milan (p = 0.034) and UCSF (p = 0.012), a greater amount of nodules (p = 0.047) and larger tumor burden (p = 0.005) had worse RFS. For non-cirrhotic patients, OS and RFS at 1, 3 and 5 years were 81%, 65%, 60% and 66%, 46%, 41% respectively. A greater number of nodules (p = 0.025) was associated with poorer OS.
Conclusions: Our study shows that liver resection is a safe procedure and can produce excellent results, both in cirrhotic and non-cirrhotic patients, independently to the size and number of tumors.
Introduction
Hepatocellular carcinoma (HCC) remains to date the most common primary tumor of the liver, and the sixth most frequent tumor worldwide [1]. 90% of all HCC thrive in the context of a pathological liver, usually due to hepatitis B virus (HBV), hepatitis C virus (HCV), or alcohol abuse. Universal vaccination against HBV, new effective antiretroviral therapies against HCV and lifestyle changes towards a sedentary, obese general population with metabolic syndrome are soon to tip the scale towards other causes of cirrhosis, making non alcoholic steatohepatitis (NASH) the leading cause of HCC in western countries [2,3].
HCC can, although rarely, present itself in a non-cirrhotic liver. In these patients, physiopathological and epidemiological factors (hereditary diseases, genomic mutations and exposure to external factors such as hormones and agrochemical products) greatly differ from the ones associated to cirrrhotic liver-HCC, and should therefore be considered a different clinical entity.
Therapeutic options in HCC differ according to the context upon which it settles. For healthy, non-cirrhotic livers, resective surgery remains the best alternative with very acceptable results, whereas HCC in a cirrhotic liver presents itself as a true challenge for physicians and surgeons alike. The Barcelona Clinic Liver Cancer (BCLC) [4] staging system is currently accepted worldwide as a guideline for decision taking in HCC, and is used in several society guidelines. BCLC staging stratifies patients according to Milan criteria [5], liver function (Child-Pugh-Turcotte score) and the patient's general clinical condition in five categories (very early-early-intermediate-advanced-terminal). This aids the physician in decision making for each individual patient. Liver resection is usually limited to small tumors with good liver function (BCLC 0-A), in patients without portal hypertension.
The aim of this study was to report and analyze the results of resected HCC in both cirrhotic and non-cirrhotic patients in our institution in a 12 year period. Short term postoperative outcomes as well as long term outcomes such as overall survival (OS) and recurrence-free survival (RFS) are analyzed and presented, as well as the possible factors that may influence survival.
Methods
Patients
The population of this study consisted of patients with HCC who underwent surgical resection as an intent-to-cure therapy by the hepato-pancreato-biliary section of our institution between January 2007 and december 2018. All data was retrospectively recollected using our institutional electronic medical records and stored in a remote database.
This study was approved by the institutional Ethics Committee (Protocol number 3474), and is in consonance with the Helsinki Declaration.
Patients with a history of previous liver resection were excluded, so were patients who had received previous treatments with intent-to-cure such as radiofrequency ablation (RFA) or orthotopic liver transplantation (OLT). Patients with missing data in medical records were also excluded. Medical background and physical examination was initially performed. Afterwards, serological tests were performed, which included basic blood cell counts, renal and liver function tests, such as creatinine, bilirubin, prothrombin time, HBV and HCV antigen and antibody loads. Imaging studies included chest X-ray, abdominal ultrasound, CT scan and magnetic resonance. Cirrhotic patients were surveilled for varices with gastroscopy, and liver fibrosis was assessed by biopsy, fibroscan or evidence of portal hypertension as a surrogate.
Definitions
Preoperative diagnosis of HCC was based mainly on radiological findings, as well as anatomopathological assessment. LI-RADS system [6] was used to diagnose HCC in at least one high quality method (contrast-enhanced abdominal CT scan, or gadolinium-enhanced abdominal magnetic resonance). Those cases without radiological certainty were defined via percutaneous biopsy. Cirrhosis was diagnosed either by liver biopsy or fibroscan using METAVIR scale [7], or by evidence of extrahepatic manifestations such as esophageal varices. Postoperative complications were documented and analysed using the Dindo Clavien grading system [8], grouping grades I and II as minor complications and III-V major complications. Postoperative liver failure (the inability of the liver to correctly synthesize and excrete metabolites) was defined and graded using the ISGLS definition [9] of elevated INR and Bilirubin levels at day 5 or more. Overall survival was defined as the time lapse between the liver resection and patient death or loss of follow-up, whichever the cause. Recurrence free survival as the time lapse between surgical treatment and the apparition of radiological evidence suggestive of HCC.
Statistical analysis
Data was analysed using STATA MP -13 (Statacorp LLC, Texas, EEUU). Numerical data is shown as a mean and standard deviation (SD), or median and range when appropriate. Categorical variables were compared using χ2 or Fisher's exact test when appropriate, and continuous variables were compared using the independent sample t-test. A p value of ≤ 0.05 was considered statistically significant, with a 95% confidence interval. OS and RFS were obtained via Kaplan-Meier curves, compared with log-rank tests. Cox regression model was used for univariate and multivariate analysis.
Results
A total of 93 patients were submitted for surgical resection of HCC at our center between 2007 and 2018. Of these 93 patients, 86 met all of our inclusion and none of the exclusion criteria. Clinical features, radiological findings, preoperative data and specimen findings are shown in Table 1. Of all 86 patients, 49 presented in the setting of a cirrhotic liver, while 37 patients had HCC in a non-cirrhotic liver. Age was similar in both groups, with a mean of 64.02 years (SD 12.56) in the cirrhotic group, and 60.32 (SD 17.97) in the non-cirrhotic cohort.
Both groups had a male predominance, 71% in the cirrhotic group and 60% in the non-cirrhotic group. Results were analyzed for both cohorts separately.
Cirrhotic patients
The vast majority of HCC were diagnosed either by sonographical surveillance (36%), or presented with nonspecific symptoms (30%). At diagnosis, the largest tumor size on average was 68.26 mm, with an average of 1.95 nodules per patient. Eight patients (16.32%) had macrovascular invasion at diagnosis. Interestingly, only 38% of patients underwent surgery within Milan Criteria (19 patients), and 46.94% within UCSF criteria (23 patients). Specimen data are also included in Table 1.
The most frequent procedures were atypical hepatectomies accounting for 40.82% of all procedures (20 patients). Right hepatectomy was second in frequency, performed in 10 patients. A total of 30 patients received a minor hepatectomy (3 sections or less), with the remaining 19 patients being submitted to major hepatectomy. Preoperatory portal embolization was necessary in 11 patients (20%) (Table 2).
Tabla 2: Surgical procedures. View Table 2
Postoperative complications are summarized in Table 3. A total of 25 complications were recorded in these 49 patients. Most of them consisted of minor complications (18 complications). Five major complications were recorded for this cohort. Two patients required postoperative pleural drainage, one due to pneumothorax, the other to pleural effusion. A third patient required CT-guided percutaneous drainage of a postoperative biloma. One patient was reoperated due to evisceration. Two of the 49 patients (4%) died in the early postoperative period. The first one had to be intervened immediately after original surgery due to hypovolemic shock secondary to hemorrhage, and evolved torpidly afterwards. The second patient developed postoperative liver failure followed by multiorgan failure. The subject had undergone a right hepatectomy without prior portal embolization.
Table 3: Postoperative complications using Dindo-Clavien grading system. View Table 3
The mean follow-up time was 31.50 months (0.5 months-102 months). OS for the cirrhotic patients at 1, 3 and 5 years was 77%, 59%, 49% respectively. RFS at 1, 3 and 5 years for this group was 75.8%, 43.9%, 25%. RFS and OS Kapplan-Meier curves are shown in Figure 1 and Figure 2. Table 4 shows univariate analysis of different clinical, radiological and specimen factors regarding OS and RFS. Patients outside UCSF criteria (p = 0.05), several nodules in the specimen (p = 0.004) and a larger total tumor size (sum of the largest diameter of each nodule) with a (p = 0.02) were all associated with poorer overall survival. Regarding RFS, patients outside Milan (p = 0.034) and UCSF (p = 0.012) criteria, a greater amount of nodules (p = 0.047) and a larger tumor burden (p = 0.005) were all associated with worse outcomes. Multivariate analysis for these variables can be seen in Table 5. A greater tumor number (p = 0.025) was the only independent factor associated with poorer OS, while there was no independent factor linked to RFS in the multivariate analysis.
Table 4: Univariate analyses for OS and RFS for Liver resection. View Table 4
Table 5: Multivariate analysis for OS and RFS in Cirrhotic patients. View Table 5
Non-Cirrhotic patients
54% of patients presented with nonspecific symptoms which led to the diagnosis, and 32.43% of all 37 HCC presented as incidentalomas. Largest tumor at diagnosis had a mean value of 110 mm, with an average of 1.24 nodules per patient. 24% (9 patients) presented macrovascular invasion as a radiological finding. 8 patients were operated within UCSF criteria, and only 5 (13.5%) within Milan. Baseline characteristics can be seen in Table 1.
Only 32.4% (12 patients) underwent minor liver resections (3 or less sections). The rest underwent proper right hepatectomy (12 patients), or greater (extended hepatectomy or Trisectionectomy). 6 patients underwent portal embolization prior to a greater hepatectomy. Surgical procedures are shown in Table 2.
Table 3 shows complications graded by Dindo-Clavien. A total of 14 complications were observed, of which 9 (64.2%) were minor complications. 3 patients required image-guided percutaneous drainage of intraabdominal abscesses. 1 patient required reintervention due to wound disruption. 1 patient evolved with clinically significant biliary leak secondary to anastomotic disruption early on after the initial surgery. The subject was intervened on and the disruption repaired, but died due to organ failure and sepsis.
Median follow up time was 32.13 months (range 0.3-105 months). OS and RFS at 1, 3 and 5 years for the non-cirrhotic cohort were 81%, 65%, 60% and 66%, 46%, 41% respectively. Kapplan Meier curves for this group are also shown in Figure 1 and Figure 2. Univariate analysis for several variables is also shown in Table 4. A greater number of nodules (p = 0.025) proved itself to be a factor associated with poorer OS. Univariate analysis for RFS could not demonstrate any risk factors for our non-cirrhotic cohort.
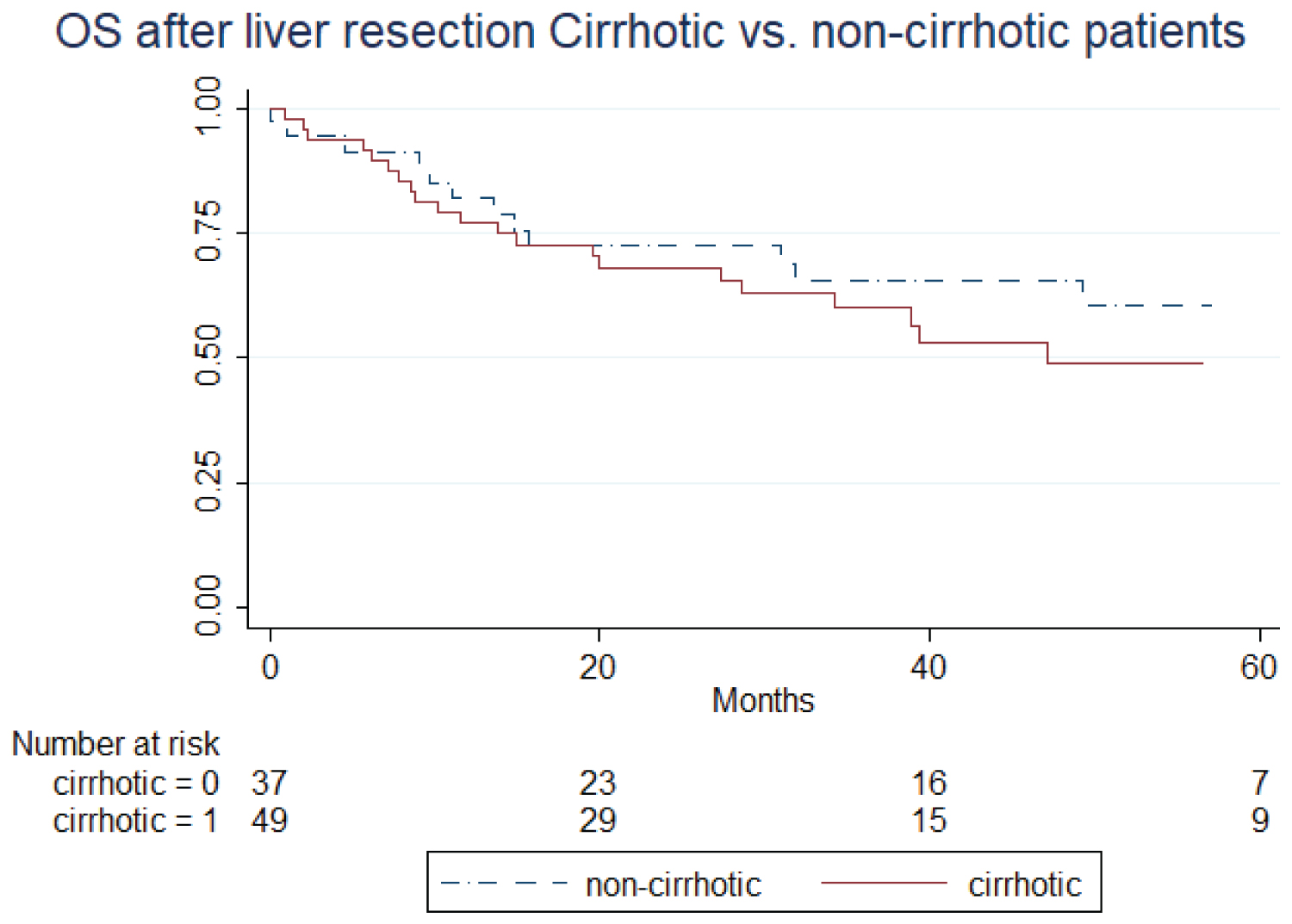
Figure 1: Overall survival after liver resection in cirrhotic and non-cirrhotic patients. Kaplan-Meier curve.
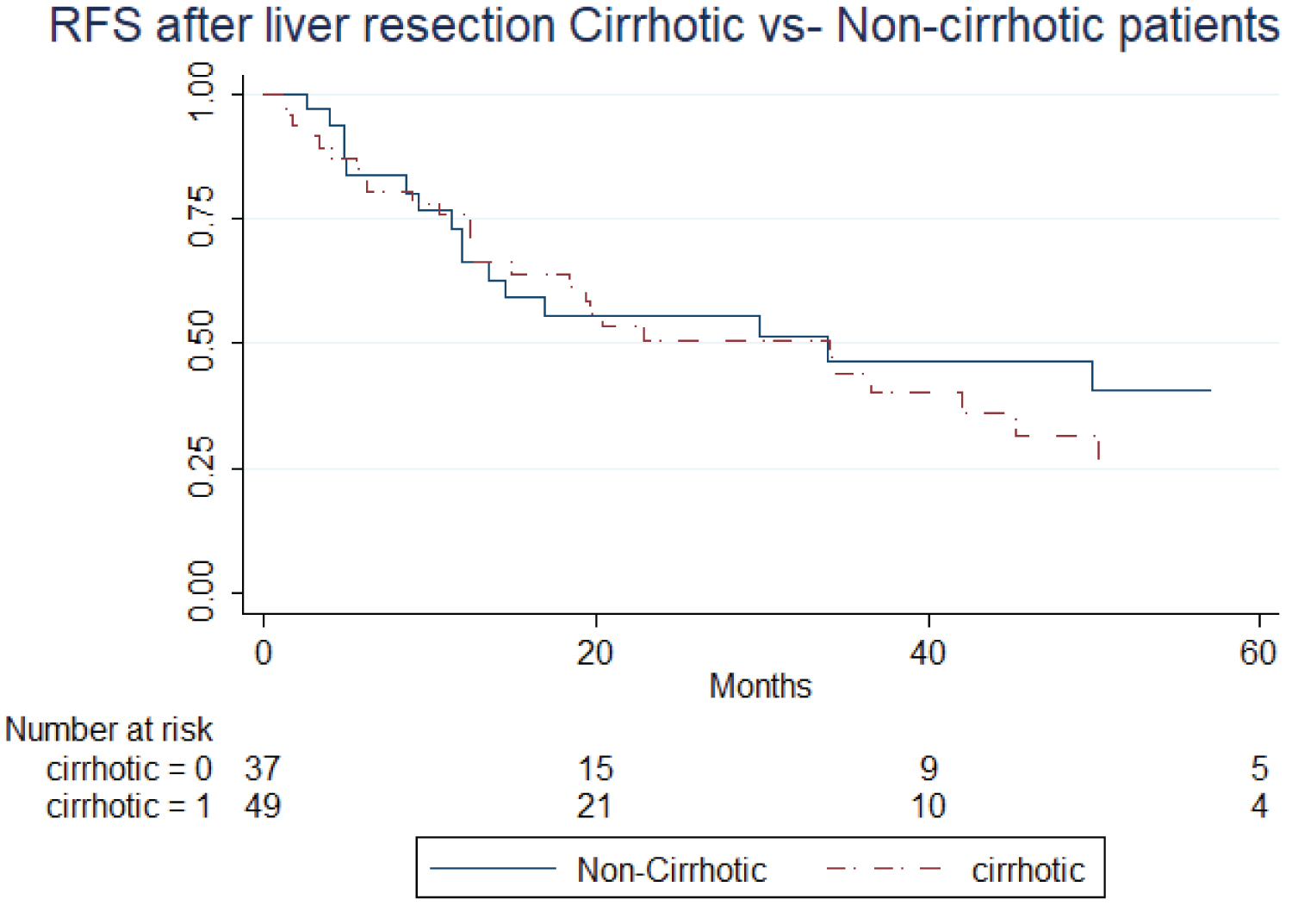
Figure 2: Recurrence-free survival after liver resection in cirrhotic and non-cirrhotic patients. Kaplan-Meier curve.
Discussion
HCC is an extremely complex entity, from its presentation in patients with a crippling disease such as cirrhosis to its oligosymptomatic presentation in non-cirrhotic population. Only 30% of HCC patients are suitable candidates for curative treatment on diagnosis, usually due to multicentricity [10]. Considering these numbers, early detection by effective screening programs is a crucial element in management of HCC. BCLC staging system is accepted worldwide as the benchmark for HCC staging in cirrhotic patients. Inside BCLC, curative resection is considered for patients within Milan criteria, with incipient liver disease (Child-Pugh A/super A), without portal hypertension or significant liver impairment (Bilirubin levels), and has a OS and RFS comparable to OLT [11].
This study analyses 86 patients who underwent surgical resection, divided according to the presence of cirrhosis, in a time lapse of 12 years in our institution. On average, the largest tumor size for the cirrhotic cohort on the specimen 59.38 mm, and 97.56 mm for the non-cirrhotic cohort. This reflects the asymptomatic presentation in the non-cirrhotic population, as well as screening techniques in cirrhotic population. Long term outcomes regarding OS and RFS for our center are comparable to other current series [12-16]. Grigoire, et al. [16] published a series of 844 patients with HCC, divided into three groups with an intent-to-cure treatment: OLT, Liver resection and RFA. 518 of these patients were surgically resected, with OS at 1, 3 and 5 years of 82, 55 and 37%. These results are quite similar to our cohort, which presents a better 5 year OS (49%). It should be noted that most patients presented by Grigoire, et al. had single nodules (86%), and 55% of them were resected within Milan criteria, whereas only 38% of our patients were intervened on within Milan. Menahen, et al. [17] performed a meta-analysis of nine studies involving a total of 570 and 861 patients who underwent liver resection and OLT respectively. The OS for the liver resection group at 1 and 5 years respectively were 84.5% and 47.9%. RFS at 1, 3 and 5 years was available for 419 of liver resection patients, and were as follows: 83%, 54.4% and 35.6%. The study found no difference in OS at 1, 3 or 5 years, but did find a significant improvement at 10 years. RFS was significantly better at 3 and 5 years for the OLT group. It is to be duly noted once again that these survival rates (very similar to the ones in our series), are restricted to patients within Milan criteria, whereas our cohort counts with an important number of resected patients outside Milan criteria. Eligibility for liver resection has been one of the main debates regarding BCLC classification, being criticized by many for being too strict. In the last years several surgical groups aim to prove the validity of surgical resection beyond BCLC [18,19]. There are several studies that aim to validate liver resection for patients outside Milan/BCLC criteria. Last year, Tsilimigras, et al. [15] published a multicenter study of liver resections for HCC within and without BCLC. Although OS and RFS at 1, 3 and 5 years were significantly worse in the BCLC-B/C group, results were acceptable, but further prospective studies matching liver resection with adequate therapies (TACE or systemic therapies) are needed. Mortality for our cirrhotic cohort was 4%, in consonance with EASL-EORTC international guidelines [20].
Our non-cirrhotic group presented slightly better results in terms of OS and RFS when compared to the cirrhotic group. This is not a new finding, as it has been described by numerous other studies, including a 51 patient cohort from our own experience in 2010 [21]. This cohort of patients selected from 1990 to 2006 had a short term follow up of 1 2 and 3 years with a 3 years OS of 67% and a RFS of 37%, quite similar to our current study (65% OS and 46% RFS). Likewise, the mean tumoral burden for Ardiles, et al. study was of 80 mm, larger than the usual HCC in cirrhotic patients, but smaller than our current tumor size (110 mm). This most probably reflects the effects of experience and new techniques (portal ligation, ALLPS) on patient eligibility for major hepatic procedures.
There are but a few latin american studies regarding resected HCC. In 2007, Ruiz, et al. [22] published a series of 232 liver resections in HCC patients. Of the 190 R0 resections, only 31 were set in cirrhotic livers, and average tumor size was 15 mm, with AFP levels of 5467 ng/dl, for an average age of merely 36 years. These findings are in consonance with the high number of HCC in non-cirrhotic patients in this study. Despite the relatively high number of subjects included in the study (in a time lapse of over 25 years), lack of information regarding Milan, UCSF and BCLC stages for cirrhotic patients make it difficult to compare results to this series, and may possibly account for the lower OS and RFS in this study compared to the authors'. In 2010, Yañes, et al. [23] presented a series of 38 patients who underwent surgical treatment. This included OLT, RFA and LR. Only 8 patients underwent LR, with a 3 year OS of 85.7% and no recurrence. No additional demographic data or information regarding size, staging and cirrhosis are shown. The small number of patients and the lack of data greatly hinders any attempt at comparison. Martinez-Mier, et al. [24] recently published their findings regarding preoperative AFP levels and surgical outcomes in HCC. 19 BCLC A patients were included in this study, with a AFP average level of 463 ng/dl. Although no RFS data is presented, OS at 5 years was 55.9%. The authors of this study concluded that AFP levels over 15 ng/dl proved to be an independent factor for poorer OS in surgically resected patients.
The authors believe our series, although not quite as large in numbers as other studies worldwide, is one of the most complete and well designed studies for LR in HCC in Latin America, and compares very well to several series in other centers worldwide, showing that surgical resection is a valid alternative in developing countries as well.
Our study has its limitations. As a retrospective study, we encountered information bias when attempting to retrieve information from the medical records. This is specially the case for earlier patients (2007-2010). Another limitation to our study is follow-up drop out. Being a referral center for our country, it is not rare to find that many patients come to our institution for the surgical intervention, and continue their follow up in their hometown. We attempted to minimize the follow up loss by contacting lost patients to their last registered contact. Those patients who had recurrence and then lost follow up were assumed deceased, in order to reduce follow up bias.
Conclusions
Numerous studies prove liver resection to be a valid alternative to OLT in selected patients in terms of OS and RFS. This being said, OLT still presents superior results in the long term [17]. This is due to the fact OLT treats not only HCC, but the underlying liver disease, reducing the risk of multicentricity. Nonetheless, liver resection becomes a useful tool in the surgeon's arsenal in donor-scarce regions such as our country. For non-cirrhotic HCC, liver resection remains the standard of care as a curable treatment, with excellent results. Our study shows that liver resection is a safe procedure and can produce excellent results, both in cirrhotic and non-cirrhotic patients, independently to the size and number of tumors.
Acknowledgements
None.
Conflicts of Interest and Source of Funding
None of the authors of this manuscript has any direct or indirect commercial financial incentive associated with the publication of this paper. The funding involved in this work has been provided by the National Cancer Institute of Argentina.
References
- https://gco.iarc.fr/today/data/factsheets/populations/900-world-fact-sheets.pdf
- Trevisani F, Frigerio M, Santi V, et al. (2010) Hepatocellular carcinoma in non-cirrhotic liver: A reappraisal. Dig Liver Dis 42: 341-347.
- Nzeako UC (1995) Etiologic factors and clinical presentation of hepatocellular carcinoma: Differences between cirrhotic and noncirrhotic Italian patients. Cancer 76: 915.
- Forner A, Llovet JM, Bruix J (2012) Hepatocellular carcinoma. Lancet 379: 1245-1255.
- Mazzaferro V, Regalia E, Doci R, et al. (1996) Liver transplantation for the treatment of small hepatocellular carcinomas in patients with cirrhosis. N Engl J Med 334: 693-699.
- Mitchell DG, Bruix J, Sherman M, et al. (2015) LI-RADS (Liver Imaging Reporting and Data System): summary, discussion, and consensus of the LI-RADS Management Working Group and future directions. Hepatology 61: 1056-1065.
- Bedossa P, Poynard T (1996) An algorithm for the grading of activity in chronic hepatitis C [Internet]. Hepatology 24: 289-293.
- Dindo D, Demartines N, Clavien P-A (2004) Classification of surgical complications: A new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 240: 205-213.
- Rahbari NN, Garden OJ, Padbury R, et al. (2011) Posthepatectomy liver failure: A definition and grading by the International Study Group of Liver Surgery (ISGLS). Surgery 149: 713-724.
- Belghiti J (2008) Surgical Treatment [Internet]. Hepatocellular Carcinoma 387-408.
- Forner A, Reig M, Bruix J (2018) Hepatocellular carcinoma. Lancet 391: 1301-1314.
- Bhandare MS, Patkar S, Shetty N, et al. (2018) Liver resection for HCC outside the BCLC criteria. Langenbecks Arch Surg 403: 37-44.
- Lim C, Salloum C, Osseis M, et al. (2018) Short-term outcomes following hepatectomy for hepatocellular carcinoma within and beyond the BCLC guidelines: A prospective study HPB 20: 222-230.
- Hyun MH, Lee YS, Kim JH, et al. (2018) Hepatic resection compared to chemoembolization in intermediate- to advanced-stage hepatocellular carcinoma: A meta-analysis of high-quality studies. Hepatology 68: 977-993.
- Tsilimigras DI, Bagante F, Moris D, et al. (2020) Recurrence Patterns and Outcomes after Resection of Hepatocellular Carcinoma within and beyond the Barcelona Clinic Liver Cancer Criteria. Ann Surg Oncol 27: 2321-2331.
- Grigorie R, Alexandrescu S, Smira G, et al. (2017) Curative Intent Treatment of Hepatocellular Carcinoma - 844 Cases Treated in a General Surgery and Liver Transplantation Center [Internet]. Chirurgia 112: 289-300.
- Menahem B, Lubrano J, Duvoux C, et al. (2017) Liver transplantation versus liver resection for hepatocellular carcinoma in intention to treat: An attempt to perform an ideal meta-analysis. Liver Transpl 23: 836-844.
- Wada H, Eguchi H, Noda T, et al. (2016) Selection criteria for hepatic resection in intermediate-stage (BCLC stage B) multiple hepatocellular carcinoma [Internet]. Surgery 160: 1227-1235.
- Tsilimigras DI, Bagante F, Moris D, et al. (2019) Defining the chance of cure after resection for hepatocellular carcinoma within and beyond the Barcelona Clinic Liver Cancer guidelines: A multi-institutional analysis of 1,010 patients. Surgery 166: 967-974.
- Liver EAFTS of T (2012) European Association for the Study of the Liver, European Organisation for Research and Treatment of Cancer. EASL–EORTC Clinical Practice Guidelines: Management of hepatocellular carcinoma [Internet]. Journal of Hepatology 56: 908-943.
- Ardiles V, Sánchez Clariá R, Mazza OM, et al. (2010) Prognostic factors after resection of hepatocellular carcinoma in the non-cirrhotic liver: presentation of 51 cases. Cir Esp 87: 148-154.
- Ruiz E, Sanchez J, Celis J, et al. (2007) Resultados a corto y largo plazo de la Resección Hepática por Hepatocarcinoma. Análisis de 232 Resecciones Consecutivas. Rev Gastroenterol Perú 27: 223-235.
- Yáñez MR, Gamboa CC, Weisse AO, et al. (2010) Tratamiento quirúrgico en hepatocarcinoma: Experiencia preliminar [Internet]. Revista chilena de cirugía 62: 22-26.
- Martínez-Mier G, Esquivel-Torres S, Nava-Lacorte A, et al. (2017) Correlación de los niveles de alfafetoproteína sérica preoperatoria y sobrevida en el tratamiento quirúrgico del hepatocarcinoma en una unidad médica de alta especialidad en Veracruz, México [Internet]. Revista de Gastroenterología de México 82: 357-360.
INDEXING
PARTNERS









Table 1: Baseline characteristics. View Table 1