-
Address:
17888 67th Court North
Loxahatchee, FL
-
Mail us:
contact@wrightacademia.org
- submit manuscript
Table of content
Case Report |
Open Access |
Volume 2 | Issue 1 |
Aetiology and Management of Acute Thrombocytopenia Post PCI: A Case Report
Prakash Sanzgiri, Charan Reddy KV, Srinivas Kudwa and Rohan Thanedar
Department of Clinical and Interventional Cardiology, Lilavati Hospital and Research Centre, India
*Corresponding author: Charan Reddy KV, Department of Clinical and Interventional Cardiology, Lilavati Hospital and Research Centre, Bandra (W), Mumbai-50, India, E-mail: chrnr@rediffmail.com
Citation: Sanzgiri P, Charan Reddy KV, Kudwa S, Thanedar R (2019) Aetiology and Management of Acute Thrombocytopenia Post PCI: A Case Report. Cardiol Cases Syst Rev. 2:006.
Copyright © Sanzgiri P, et al.
Received: |
Accepted: |
Published: |
Thrombocytopenia (TP) after Percutaneous Coronary Intervention (PCI) is not uncommon and can be associated with increased morbidity and mortality. TP is a recognized complication of treatment with Glycoprotein GPIIb/IIIa inhibitors (GPIs) inhibitors in Acute Coronary Syndrome (ACS). However, majority of GPI-induced TPcases can be managed conservatively with only a few requiring more active intervention. The specific treatment plan generally depends on the underlying aetiology of TP. Here, we present a case of acute TP after eptifibatide treatment following primary PCI and discuss various causes, diagnostic modalities and treatment options.
Introduction
Endothelial damage and associated plaque rupture exposes various thrombogenic components to blood, inducing platelet activation and platelet aggregation that are hallmarks of Acute Coronary Syndrome (ACS) [1]. Percutaneous Coronary Intervention (PCI) with drug coated stents improves epicardial blood flow and is a validated therapeutic option [2]. However, the stent struts and the exposed subendocardial surface of coronary arteries are also thrombogenic; hence anti-platelet drugs are given to prevent immediate stent thrombosis [3]. The efficacy of Glycoprotein IIb/IIIa antagonists (GPI's) in the treatment of acute coronary syndromes (unstable angina and non-Q wave MI), MI and PCI has been validated in many multi-centric trials [4,5]. Here, we describe a case of acute thrombocytopenia after a 12 hr administration of eptifibatide following primary PCI for acute inferior wall STEMI. Here we present such a case of acute thrombocytopenia and discuss the various causes, diagnostic modalities and treatment options.
Case Report
A 76-year male, with no history of Diabetes Mellitus (DM) or hypertension, presented to the emergency room with sudden onset retrosternal chest pain and sweating for the past one hour. Blood pressure and chest auscultation were normal. ECG revealed ST segment elevation in the Inferior leads (II, III and VF with reciprocal changes in the anterior leads) (Figure 1). Oral aspirin (325 mg), clopidogrel (600 mg) and atorvastatin (80 mg) were given. 2-D Echowas done, which showed regional wall motion abnormality in inferior wall and inferior-septal segments with preserved wall thickness and LV ejection fraction was 45%. RV function was normal and no valve dysfunction observed. Patient was immediately shifted to cardiac catheterization laboratory for primary angioplasty.
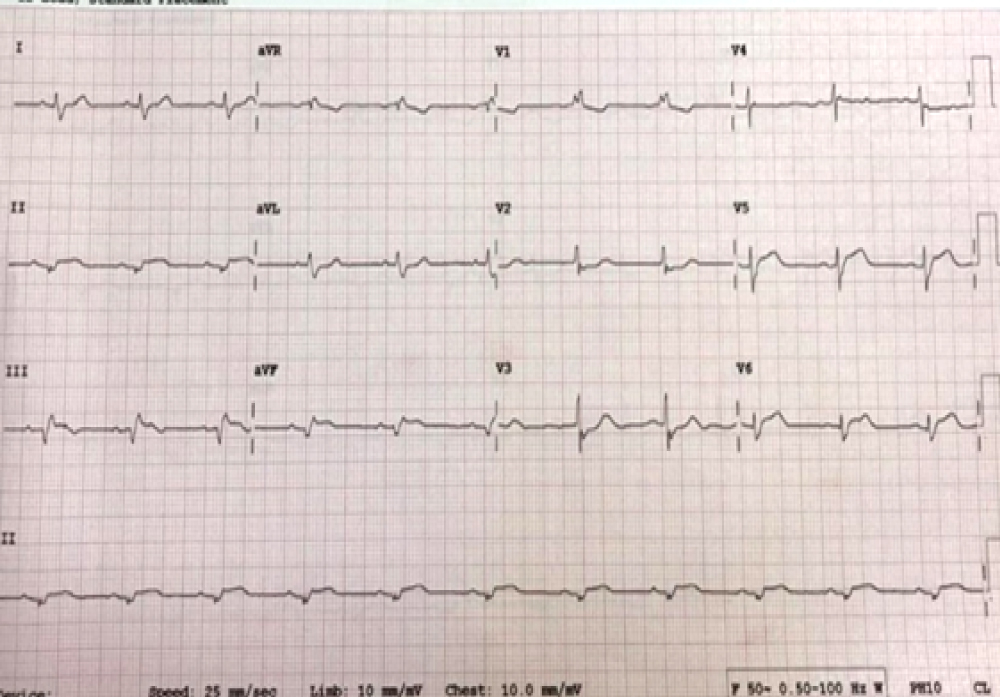
Figure 1: ECG showing Acute Inferior wall STEMI (lead II, III and aVF) with reciprocal changes in anterior leads (V2, V3, V4).
Coronary angiography showed a 90% thrombotic occlusion in dominant Right Coronary Artery (RCA) with thrombolysis in myocardial infarction-2 (TIMI-2) flow (Figure 2). Proximal and mid segment of the Left Anterior Descending (LAD) also had two atherosclerotic lesions of 80% and 70% stenosis respectively. Non-dominant Circumflex artery was normal. After appropriate weight adjusted heparinization, Percutaneous Transluminal Coronary Angioplasty (PTCA) was planned as a staged procedure. Thrombus aspiration of RCA was done with a 6F Export catheter. However this did not establish adequate antegrade flow. Bolus and maintenance infusion (weight adjusted) of eptifibatide (GpIIb/IIIa inhibitor) was given, to be continued for 12 hrs. RCA was then directly stented with an Ultimaster sirolimus drug-eluting stent of size 3 × 15 mm. TIMI-3 flow was established. Patient had relief in symptoms; ECG showed good ST segment resolution, no arrhythmias with normal hemodynamical parameters. Door to Balloon Time (DBT) was 34 minutes. He was later shifted to the cardiac Care Unit for observation. Plan was to perform angioplasty to the Left anterior descending (LAD) lesion before the present discharge.

Figure 2: a) CAG showing Thrombotic occlusion (white arrow) of RCA and; b) Two atherosclerotic lesions of 80% and 70% stenosis in the LAD.
However, complete blood count performed after 12 hr showed a severe drop in platelet count from 2,54,000 (pre-procedure) to 4,000 (post-procedure). Since, the patient was asymptomatic with no clinical evidence of bleeding, he was managed conservatively. To rule out Heparin-Induced Thrombocytopenia (HIT), antibody test was done, which was negative. Schistocytes were absent in the peripheral smear. Close observation and repeat platelet count was performed (Table 1). Platelet counts showed gradual improvement after 48 hours. Aspirin (75 mg) and clopidogrel (150 mg) were continued. He was discharged on the 7th day, readmitted after 4 weeks and successfully underwent angioplasty to the LAD without any complications.
Discussion
The standard definition of thrombocytopenia is an absolute reduction in platelet count to < 150,000/mm3. Acute thrombocytopenia is defined as a platelet count < 100,000/mm3, acute severe thrombocytopenia is defined as platelet count < 50,000/mm3, and acute profound thrombocytopenia as platelet count < 20,000/mm3 within 24 hrs of the exposure [6,7].
Thrombocytopenia is not infrequent in patients following PCI, since; various therapies and mechanical devices used can potentially cause a decrease in the platelet counts. Identification of the cause is critical because management varies significantly based on the aetiology. Differential diagnosis of severe thrombocytopenia post PCI is as follows.
Laboratory pseudo-thrombocytopenia
It is an in-vivo artefact resulting from agglutination of platelets. It is a benign condition of no clinical significance. It is caused by platelet aggregation when exposed to Ethylene Diamine Tetra-Acetic Acid (EDTA) in the specimen tube or low temperatures. The diagnosis is confirmed by examining platelet clumping in peripheral blood film or using a citrate-containing specimen tube for blood collection, which does not cause platelet aggregation [7].
Platelet glycoprotein IIb/IIIa inhibitors (GPIs) induced thrombocytopenia
The GPIIb/IIIa antagonists occupy fibrinogen binding site at the GP IIb/IIIa, blocking platelet aggregation. There are two categories of GPIIb/IIIa inhibitors: Drugs that exhibit avid receptor binding (abciximab) and drugs which have competitive, dose-dependent pharmaco dynamics (eptifibatide and tirofiban). Abciximab is long-acting with biologic half-life (6 to 12 hrs). Even after 24 hrs, abciximab still has 50% to 60% platelet blockade. The plasma half-life of eptifibatide is shorter at 2.5 hrs. At 4 hrs after stopping eptifibatide, platelet aggregation is approximately 70% of normal, which promotes early return to normal haemostasis, thereby, reducing bleeding complications [6,8-10].
Abciximab, eptifibatide and tirofiban can cause thrombocytopenia to a variable extent. (1-2% with abciximab and 0.5-1.2% with eptifibatide and tirofiban). However a second exposure of the drug in susceptible patients can cause more profound drop in platelet counts. GPI-related thrombocytopenia is immune-mediated and occurs rapidly within a few hours after the administration of the drug due to the presence of naturally occurring drug dependent anti-platelet antibodies. Unlike Heparin Induced Thrombocytopenia (HIT), GPIIb/IIIa inhibitor associated thrombocytopenia can be rapid, more profound, with very low platelet count. In post-PCI patients treated with heparin and a GPIIb/IIIa inhibitor, an abrupt decrease in platelet count is almost always due to the GPIIb/IIIa inhibitor rather than to HIT.
In a meta-analysis of 29 randomized large trials (> 1000 patients) of GPI vs. placebo involving a total of 123,419 patients, the administration of GPI compared with placebo was associated with a 63% increased risk of thrombocytopenia (< 100,000 platelets/mm3), and > 3-fold increased risk of severe thrombocytopenia (< 50,000 platelets/mm3) [5].
The anti-platelet effects of both eptifibatide and tirofiban are dose dependent. Therefore, reversal of the effect requires discontinuation of the respective agents to give time for body clearance. Thrombocytopenia caused by eptifibatide or tirofiban, resolves with time, as both drugs have a short half-life. Platelet transfusions may be given only to asymptomatic patients on abciximab with very low platelet counts (< 20,000 platelets/mm3) or for patients with severe uncontrolled bleeding, or that undergoing early bypass surgery with a platelet count of < 50,000 platelets/mm3.
Heparin induced thrombocytopenia (HIT)
Heparin Induced Thrombocytopenia (HIT) is caused by antibodies directed to complexes containing heparin and an endogenous platelet protein, platelet factor-4 (PF4). The prevalence of HIT generally varies from 0.5% to 5% of patients, depending on the population studied, the type of heparin used, and the duration of heparin therapy. The risk of HIT is lower with LMWH compared to UFH [6,11].
About 8% of heparinised patients have antibodies without symptoms, and 1-5% have thrombocytopenia. About 1/3 of these develop arterial or venous thrombosis with 20-30% mortality. In most patients, thrombotic complications occur concurrently with thrombocytopenia.
To prevent HIT, avoid the use of heparin, but need alternative anticoagulant to prevent life threatening thrombo-embolic complications (e.g. skin necrosis, gangrene of the extremities, myocardial infarction, pulmonary embolism, and stroke). Enoxaparin may cross react with heparin antibodies causing thrombosis. Fond aparinux or direct thrombin inhibitor like bivalirudin or NOACs (rivaroxaban, apixaban, or dabigatran) can be used.
Thienopyridine derivative inducing thrombocytopenic purpura
The thienopyridine derivatives (ticlopidine, clopidogrel, and prasugrel) are known to be associated with Thrombotic Thrombocytopenic Purpura (TTP). Recently, few cases of ticagrelor (a non-thienopyridine derivative) treated patients developing TTP has also been reported [12].
TTP and Haemolytic Uremic Syndrome (HUS) are similar disorders classified under Thrombotic Microangiopathy (TMA) due to the common mechanism of platelet and Red Blood Cells (RBC) destruction in the microvasculature. Both TTP and HUS are characterized by the deposition of platelets and fibrin in small vessels, causing mechanical shearing of RBCs and agglutination of platelets and resulting in Micro-Angiopathic Haemolytic Anaemia (MAHA) and thrombocytopenia with presence of schistocytes in peripheral smear. Pathogenesis of TTP was not known for over half a century until the discovery of ADAMTS-13 (A Disintegrin and Metalloproteinase with a thrombospondin type-1 motif, member-13), a zinc-containing metalloprotease enzyme, synthesized in the liver that cleaves von Willebrand factor (vWf), a large protein involved in blood clotting. TTP was found to be associated with deficiency of plasma ADAMTS-13 activity. Lack of ADAMTS-13 activity leads to accumulation of ultra-large vWF particles, which bind to active platelets and form the microthrombi. Moreover, ADAMTS-13 deficiency has also shown to be a risk factor for the development of myocardial infarction, stroke, cerebral malaria, and preeclampsia [13].
The standard treatment for TTP relies upon therapeutic Plasma Exchange (PLEX), which removes circulating inhibitory auto antibodies. Mortality from TTP was very high (> 90%) before the advent of PLEX, but has now decreased to < 20% with PLEX treatment. Despite prompt recognition and immediate therapeutic PLEX treatment, some TTP patients fail to respond. Such patients are at high risk of early death, and rituximab therapy in this condition can be lifesaving. Replacing one thienopyridine derivative for other has not been evaluated. Drugs like cilostazol or dipyridamole have poor antiplatelet activity and are not reliable in preventing thrombosis post coronary artery stenting.
Acute contrast-induced thrombocytopenia
It is a rare event with the use of modern low osmolarity iodinated contrast media [14]. The pathophysiological mechanism that causes thrombocytopenia is immunological and cell cytotoxicity. Only few cases have been described in the literature [15]. Management includes administration of high dose corticosteroids. Platelet transfusion should only be considered during clinically relevant bleeding and when HIT has been excluded.
Intra-aortic balloon pump (IABP) and other mechanical assist device associated thrombocytopenia
It occurred in 54.5% of patients with ACS undergoing an invasive strategy [16]. The thrombocytopenia is due to mechanical destruction of circulating platelets by the repeated inflation and deflation of the Intra-Aortic Balloon Pump (IABP). Limited data for newer percutaneous ventricular assist devices such as the Impella or Tandem Heart percutaneous ventricular assist device suggest that occurrence of thrombocytopenia with these devices are also not uncommon. Longer duration of counter-pulsation with IABPs can result in lower platelet counts; hence prompt removal of balloon pump is associated with a rapid increase in platelet count [17].
Thrombocytopenia can also occur due to other drugs like ACEIs, statins, NSAIDs, antibiotics, oral hypoglycemic or secondary to sepsis. Aspirin and thienopyridine derivatives should not be discontinued unless the patient has significant life-threatening, uncontrollable bleeding associated with post-PCI thrombocytopenia. Patients who develop profound thrombocytopenia due to re-exposure are more likely to require repeated platelet transfusions to normalize the platelet count, than that patient who develops thrombocytopenia after first exposure.
Approach to thrombocytopenia post PTCA can be summarized as follows:
1. Do a baseline Complete Blood Count (CBC) in all patients prior to PTCA.
2. If platelet count < 150,000/mm3, avoid the use of GPI.
3. In every patient treated with GPI, check platelet counts after initiation of therapy. If sharp drop, eptifibatide/tirofiban infusion should be immediately stopped.
4. Platelet count after 24 hrs and 36 hours should also be checked. HIT and TTP usually manifest late.
5. Early drop in platelets without exposure to GPI, consider pseudo-thrombocytopenia.
6. Do not panic if there is profound platelet count reduction. In the absence of clinical haemorrhage or drop in haemoglobin, patient may need no active intervention.
7. Do not stop Dual Anti Platelet Therapy (DAPT) in thrombocytopenia without any clinical bleeding. Acute stent thrombosis can be life threatening.
8. Microvascular ischaemia in clopidogrel induced TTP has adverse outcomes. Early detection, diagnosis and treatment can limit myocardial injury. PLEX can also reduce effect of TTP.
Conclusion
Incidence of thrombocytopenia has been reported to be 3% to 16.5% among patients with ischemic heart disease exposed to antiplatelet and antithrombin therapies [6]. This case illustrates the importance of being aware of the life threatening thrombocytopenia associated with the use of eptifibatide in ACS patient. Platelet counts before and after the initiation of GPIIb/IIIa therapy is advisable for early detection and treatment of patient before any complications.
Statement of Ethics
All the procedures followed were in accordance with the ethical standards of our Institutional ethics committee on human experimentation. Informed consent was obtained from the patient.
Funding Sources
No financial support was utilized in the preparation of this manuscript.
Disclosure Statement
The authors have no relevant conflicts of interest to report.
References
- Izak M,Bussel JB. Management of thrombocytopenia. F1000Prime Rep. 2014; 6: 45.
- Neumann FJ, Uva MS, Ahlsson A, et al. 2018 ESC/EACTS Guidelines on myocardial revascularization. European Heart J, 2019; 40: 87-165.
- Reejhsinghani R, Lotfi AS. Prevention of stent thrombosis: challenges and solutions. Vasc Health Risk Manag. 2015; 11: 93-106.
- O'Donoghue M, Antman EM, Braunwald E. The efficacy and safety of prasugrel with and without a glycoprotein IIb/IIIa inhibitor in patients with acute coronary syndromes undergoing percutaneous intervention: a TRITON-TIMI 38 (Trial to assess improvement in therapeutic outcomes by optimizing platelet inhibition with prasugrel-thrombolysis in myocardial infarction 38) analysis. J Am Coll Cardiol. 2009;54:678-685.
- Wessler JD, Giugliano RP. Risk of thrombocytopenia with glycoprotein IIb/IIIa inhibitors across drugs and patient populations: a meta-analysis of 29 large placebo-controlled randomized trials. European Heart Journal. 1: 2015; 97-106.
- . Berkowitz SD, Sane DC, Sigmon KN, Shavender JH, Harrington RA, Tcheng JE, Topol EJ, Califf RM. Occurrence and Clinical Significance of Thrombocytopenia in a Population Undergoing High-Risk Percutaneous Coronary Revascularization. J Am Coll Cardiol 1998;32:311-9.
- ShenoyC, Harjai K. Thrombocytopenia following Percutaneous Coronary Intervention. J IntervenCardiol. 2011;24:15-26.
- Tcheng JE. Clinical challenges of platelet glycoprotein IIb/IIIa receptor inhibitor therapy: Bleeding, reversal, thrombocytopenia, and retreatment. Am Heart J 2000;139:S38-45.
- Said S M, Hahn J, Schleyer E, Muller M, Fiedler G M, Buerke M, Prondzinsky R. Glycoprotein IIb/IIIa inhibitor-induced thrombocytopenia Diagnosis and treatment. Clin Res Cardiol.2007; 96:61-69.
- Aster RH, Curtis BR, Bougie DW, et al. Thrombocytopenia associated with the use of GPIIb/IIIa inhibitors: position paper of the ISTH working group on thrombocytopenia and GPIIb/IIIa inhibitors. J Thrombosis & Haemostasis. 4: 678-679.
- Arepally GM. Heparin-induced thrombocytopenia. Blood. 2017;129:2864-287.
- Wang X, Zhang S, Li L, Hua J, et al. Ticagrelor-induced thrombotic thrombocytopenic purpura A case report and review of the literature. Medicine.2018; 97:26(e11206).
- Zheng XL ADAMTS13 and von Willebrand Factor in thrombotic thrombocytopenic purpura. Annu Rev Med. 2015; 66: 211-225.
- Park M, Kim M, Park J, Cho J. Life-Threatening Thrombocytopeafollowing Intravenous Contrast Media Infusion. Yonsei Med J.2018 ; 59: 158-161.
- Gomez JMC, Francisco J. Guerrero Marquez, Luis Diaz-de la-Llera, Monica Fernandez-Quero, AgustinGuisado-Rasco, Manuel Villa-Gil-Ortega. Severe thrombocytopenia induced by iodinated contrast after coronary angiography: The use of gadolinium contrast and intravascular ultrasound as an alternative to guide percutaneous coronary intervention. Rev Port Cardiol. 2017;36:61.e1-61.e4.
- Sheng Z, Zhao Yan, Jiang Y. Intra-aortic balloon pumping and thrombocytopenia in patients with acute coronary syndrome Incidence, risk factors, and prognosis. Herz. 2018; 43: 555-564.
- Vonderheide RH, Thadhani R, KuterDJ.Association of thrombocytopenia with the use of intra-aortic balloon pumps. Am J Med. 1998; 105:27-32.
INDEXING
PARTNERS









Table 1: Blood parameters after the percutaneous transluminal coronary angioplasty (PTCA). View Table 1